What is Corrosion Resistance?
Corrosion resistance is the ability of a material to withstand chemical or electrochemical reactions that cause deterioration when exposed to environmental factors like moisture, oxygen, acids, or salts. This property determines how long materials maintain their structural integrity and functionality in corrosive environments.
Materials achieve corrosion resistance through two primary mechanisms: inherent properties of the material itself, or applied protective measures like coatings and treatments. Metals like stainless steel contain chromium that forms a self-healing oxide layer, while others require external protection to prevent degradation.
Why Corrosion Resistance Matters
The global cost of corrosion exceeds 3% of worldwide GDP annually, according to the Association for Materials Protection and Performance. Between 15-33% of annual steel production replaces corroded steel, accounting for 1.6-3.4% of global CO₂ emissions. These figures reveal corrosion as both an economic burden and environmental challenge.
Industries from aerospace to marine engineering depend on corrosion-resistant materials to prevent catastrophic failures. A corroded aircraft component or offshore oil platform can lead to safety disasters and massive financial losses. In medical devices, poor corrosion resistance can cause implant failures and patient complications. The semiconductor industry requires extremely low corrosion rates to maintain the purity and performance of manufacturing equipment.
Selecting materials with appropriate corrosion resistance reduces replacement frequency, cuts maintenance costs, and extends product lifecycles. This becomes particularly critical in applications where access for repairs is difficult or impossible, such as subsea pipelines or buried infrastructure.

How Corrosion Works
Corrosion is fundamentally an electrochemical process where metals lose electrons and convert to more stable oxide forms. When iron corrodes, for instance, it oxidizes to form iron oxide-rust. This reaction requires three elements: an anode (where oxidation occurs), a cathode (where reduction happens), and an electrolyte that conducts ions between them.
The reaction accelerates in the presence of moisture, as water acts as an electrolyte. Salt intensifies the process by increasing the solution's conductivity, which explains why marine environments are particularly aggressive. Temperature also plays a significant role-higher temperatures generally increase corrosion rates, though some materials perform better at elevated temperatures.
Different types of corrosion attack materials in distinct ways. Uniform corrosion spreads evenly across a surface, slowly thinning the material. Pitting corrosion creates deep, localized holes that can penetrate through components. Crevice corrosion occurs in tight spaces where stagnant fluid becomes trapped. Galvanic corrosion happens when dissimilar metals contact each other in an electrolyte, with the more active metal corroding preferentially.
Stress corrosion cracking combines mechanical stress with a corrosive environment to create cracks that propagate through the material. This form is particularly dangerous because external inspection may show minimal surface damage while internal cracking compromises structural integrity.
Materials and Their Corrosion Resistance
Stainless Steel
Stainless steel dominates corrosion-resistant applications due to its balance of performance and cost. The defining characteristic is a minimum 10.5% chromium content, which reacts with oxygen to form a thin chromium oxide layer on the surface. This passive layer self-heals if scratched, provided oxygen is present.
Different stainless steel grades offer varying corrosion resistance levels. Austenitic grades like 304 and 316 contain 18-20% chromium plus 8-12% nickel, providing excellent general corrosion resistance. Grade 316 adds 2-3% molybdenum, significantly improving resistance to chlorides and making it suitable for marine environments. Duplex stainless steels combine austenitic and ferritic structures, achieving higher strength with good corrosion resistance at lower cost than highly alloyed austenitic grades.
Martensitic stainless steels sacrifice some corrosion resistance for higher hardness and strength. These grades work well in applications like cutlery or surgical instruments where moderate corrosion resistance pairs with wear resistance requirements. Precipitation-hardened grades like 17-4 PH offer the best combination of strength and corrosion resistance among stainless steels, with yield strengths 50% higher than 300-series austenitic grades.
Aluminum and Its Alloys
Aluminum naturally forms a protective aluminum oxide layer when exposed to air. This layer prevents further oxidation and provides corrosion resistance in many environments. The lightweight nature and good corrosion resistance make aluminum ideal for aerospace, automotive, and marine applications.
However, not all aluminum alloys resist corrosion equally. The 2024 aluminum alloy, widely used in aerospace for its high strength-to-weight ratio, has relatively poor corrosion resistance compared to other aluminum alloys. This alloy contains 3.8-4.9% copper, which compromises corrosion resistance. Manufacturers address this by cladding 2024 aluminum with high-purity aluminum or other protective coatings, creating AlClad products that combine the strength of 2024 with improved corrosion resistance.
The 6061 aluminum alloy offers better corrosion resistance than 2024, though with lower strength. Marine-grade aluminum alloys include additional elements that enhance resistance to saltwater environments. Anodizing-an electrochemical process that artificially thickens the oxide layer-further improves aluminum's corrosion resistance while adding surface hardness.
Titanium
Titanium offers exceptional corrosion resistance through a stable oxide film that forms instantly when exposed to air or moisture. This oxide layer protects the base metal even in harsh environments like seawater, chlorine, and many acids. Titanium maintains this resistance across a wide temperature range and repairs the oxide layer automatically if damaged.
The corrosion resistance of titanium makes it valuable in chemical processing, desalination plants, and marine engineering. Medical implants use titanium extensively because it resists corrosion in physiological environments while demonstrating excellent biocompatibility. Aerospace applications leverage titanium's combination of light weight, high strength, and corrosion resistance for structural components and engine parts.
Cost represents titanium's primary limitation-it's generally the most expensive common corrosion-resistant metal. The difficulty of machining and forming titanium adds to production costs. Despite this, applications requiring long-term reliability in aggressive environments often justify the investment.
Nickel-Based Alloys
Nickel alloys deliver superior corrosion resistance in extreme conditions where stainless steels fail. Alloys like Inconel 625 and Hastelloy C-276 contain high percentages of nickel, chromium, and molybdenum, providing resistance to oxidizing and reducing acids, chlorides, and high-temperature oxidation.
These specialty alloys excel in chemical processing, oil and gas production, and power generation applications. Inconel 718 maintains mechanical properties and corrosion resistance at temperatures up to 700°C. Monel 400, a nickel-copper alloy, shows exceptional resistance to hydrofluoric acid and seawater.
The high cost of nickel-based alloys limits their use to applications where standard stainless steels cannot perform. However, their ability to operate in conditions that would rapidly destroy other materials often makes them the only viable choice for critical components in aggressive environments.
Copper and Copper Alloys
Copper forms a protective patina-a green oxidized layer-that prevents further corrosion. This characteristic makes copper suitable for plumbing, electrical applications, and architectural elements. Brass (copper-zinc) and bronze (copper-tin) alloys inherit copper's corrosion resistance while offering different mechanical properties.
Copper alloys perform well in freshwater, saltwater, and many industrial environments. They resist biofouling better than many materials, making them valuable in marine condensers and heat exchangers. The excellent thermal and electrical conductivity adds functionality beyond corrosion resistance.
Some environments attack copper aggressively. Ammonia and ammonia-containing solutions can cause stress corrosion cracking in brass. Certain sulfur compounds accelerate copper corrosion. Understanding these limitations guides proper material selection for specific applications.
Corrosion Resistance in Metal Injection Molding
Metal Injection Molding (MIM) produces complex metal components by combining metal powders with polymer binders, injection molding the mixture, then removing the binder and sintering the metal particles. This process creates near-net-shape parts with properties approaching wrought materials, making it valuable for producing intricate corrosion-resistant components.
Stainless steels dominate MIM applications due to their combination of corrosion resistance, mechanical properties, and processability. MIM 316L offers excellent corrosion resistance and strength for medical instruments, marine hardware, and industrial components. The process enables complex geometries impossible with traditional metalworking while maintaining the corrosion resistance of the base alloy.
However, MIM processing affects corrosion resistance in specific ways. During sintering, chromium can preferentially evaporate from the surface of stainless steels, creating chromium-depleted regions susceptible to pitting corrosion. Post-sintering treatments-thermal, chemical, or mechanical-restore uniform chromium distribution and recover full corrosion resistance. Surface roughness also influences corrosion behavior; polishing MIM stainless steel parts after sintering improves corrosion resistance by reducing pit initiation sites.
MIM 17-4 PH stainless steel combines high strength with good corrosion resistance for demanding applications. This precipitation-hardened grade requires proper heat treatment to achieve optimal properties. In artificial saliva tests, untreated MIM 17-4 PH can exhibit pitting, but polishing and proper processing yield corrosion resistance suitable for medical and dental applications.
Titanium MIM enables complex titanium components for aerospace and biomedical uses, though achieving full density during sintering remains challenging. The excellent corrosion resistance of titanium transfers to MIM parts when properly processed. Tool steels processed through MIM can incorporate hard phases like titanium nitride or chromium boride to enhance wear resistance while maintaining adequate corrosion protection for specific applications.
Factors Affecting Corrosion Resistance
Material composition determines baseline corrosion resistance. Alloying elements like chromium, nickel, and molybdenum form protective oxides or slow corrosion kinetics. Even small changes in composition significantly impact performance-adding 2-3% molybdenum to stainless steel dramatically improves chloride resistance.
Environmental conditions critically influence corrosion behavior. Moisture and oxygen availability drive most corrosion reactions. Salt concentration matters-seawater contains roughly 3.5% salt, while salt spray tests may use 5% solutions to accelerate testing. Temperature effects vary by material and environment; some alloys develop more protective oxide layers at elevated temperatures.
pH strongly affects corrosion rates. Acidic environments (low pH) accelerate attack on most metals, while alkaline conditions may increase or decrease corrosion depending on the material. Some metals like aluminum and zinc corrode rapidly in both acids and strong bases but resist neutral solutions.
Surface condition influences corrosion initiation. Smooth, polished surfaces provide fewer sites for pitting to begin compared to rough surfaces. Mill scale, heat treatment oxides, or contamination can create differential areas that accelerate localized corrosion. Surface treatments like passivation chemically enhance the protective oxide layer on stainless steels.
Mechanical stress contributes to stress corrosion cracking. Components under tension, especially in corrosive environments, can develop cracks that propagate until failure occurs. This form of corrosion appears in stainless steels exposed to chlorides, brass in ammonia environments, and many alloys under specific conditions. Design must consider stress levels and environmental exposure together.
Galvanic effects arise when dissimilar metals electrically contact each other in an electrolyte. The more active metal (anode) corrodes preferentially, protecting the noble metal (cathode). Material pairing and surface area ratios determine the severity-a small steel fastener in a large aluminum panel corrodes rapidly, while reversing the ratio reduces the steel's corrosion rate.
Testing and Measuring Corrosion Resistance
Corrosion resistance testing evaluates material performance under controlled conditions to predict real-world behavior. Multiple test methods exist because different applications require different assessment approaches. Results guide material selection, coating evaluation, and quality control.
Salt Spray Testing
The salt spray test (ASTM B117) is the most widely used corrosion resistance evaluation method. Metal specimens sit in a controlled chamber exposed to a continuous mist of 5% sodium chloride solution at 35°C. Test durations range from hours to thousands of hours depending on the application and expected performance.
Salt spray testing provides standardized, reproducible results that enable comparison between materials and coatings. The accelerated conditions simulate years of outdoor exposure in a fraction of the time. However, the test has limitations-it doesn't replicate cycling between wet and dry conditions that occurs in nature, potentially giving misleading results for some applications.
Variations on basic salt spray testing include copper-accelerated acetic acid salt spray (CASS) for more aggressive testing, and prohesion testing that cycles between salt spray and dry conditions to better simulate real-world exposure.
Electrochemical Testing Methods
Electrochemical techniques measure corrosion behavior by monitoring electrical properties. Linear Polarization Resistance (LPR) applies a small voltage to a sample in a corrosive solution and measures the resulting current. The polarization resistance inversely relates to corrosion rate, providing rapid, non-destructive assessment of very low corrosion rates important in pharmaceutical, food processing, and nuclear applications.
Potentiodynamic polarization scans voltage over a wide range while measuring current response. This reveals the passivation behavior of materials like stainless steel, identifying critical parameters like passive potential, passive current density, and pitting potential. Cyclic polarization helps determine susceptibility to localized corrosion.
Electrochemical Impedance Spectroscopy (EIS) applies alternating current signals at multiple frequencies, analyzing the response to characterize the protective properties of oxide films and coatings. This technique distinguishes between different corrosion mechanisms and monitors coating degradation over time.
Immersion Testing
Immersion tests submerge samples in specific corrosive solutions for defined periods. After exposure, weight loss, dimensional changes, or mechanical property degradation quantify corrosion. This method directly simulates service conditions for materials exposed to chemicals, cooling waters, or process fluids.
Exposure duration depends on the application-days for screening tests, months for qualification. Multiple samples improve statistical reliability. Visual examination, pit depth measurement, and metallographic analysis provide additional information about corrosion mechanisms.
Crevice corrosion testing uses plastic washers clamped to metal samples, creating artificial crevices where stagnant solution can cause localized attack. This evaluates susceptibility to corrosion in joints, under gaskets, and in other tight spaces.
Real-World Exposure Testing
Atmospheric exposure testing places samples in actual service environments-coastal zones, industrial areas, or specific facilities. While time-consuming, this provides the most accurate performance prediction. Controlled weathering chambers simulate outdoor conditions with UV exposure, temperature cycling, and humidity control.
Field testing reveals effects impossible to duplicate in laboratories, including biological fouling, unexpected chemical exposures, and complex interactions between multiple environmental factors. The drawback is the long duration required to generate meaningful data.
Corrosion Measurement and Rating
Corrosion rate quantifies the speed of material loss. Common units include millimeters per year (mm/yr) or mils per year (mpy), where one mil equals 0.001 inches. Rates below 0.02 mm/yr (0.8 mpy) are generally considered excellent, while rates above 0.5 mm/yr (20 mpy) indicate severe corrosion.
The formula for calculating corrosion rate from weight loss is:
Corrosion Rate = (K × W) / (A × T × D)
Where K is a constant (87.6 for mm/yr or 3.45×10⁶ for mpy), W is weight loss in grams, A is exposed area in cm², T is exposure time in hours, and D is material density in g/cm³.
Pitting factor quantifies localized corrosion severity by comparing maximum pit depth to average wall loss from uniform corrosion. Higher pitting factors indicate more concentrated attack that can cause premature failure. Pit density-number of pits per unit area-also matters for assessing risk.
Visual rating scales provide qualitative assessment. Standards define appearance criteria corresponding to different corrosion levels. While subjective, experienced inspectors achieve consistent ratings useful for coating performance comparisons.
Enhancing Corrosion Resistance
When base material corrosion resistance proves inadequate, several enhancement methods exist. The optimal approach depends on the material, environment, and application requirements.
Surface Treatments and Coatings
Protective coatings create barriers between base metal and corrosive environments. Organic coatings-paints, powder coatings, and polymer films-provide economical protection for steel structures, vehicles, and equipment. Advanced coatings incorporate self-healing mechanisms using microcapsules that release corrosion inhibitors when damaged.
Metallic coatings like zinc (galvanizing), aluminum, or zinc-aluminum alloys protect steel through both barrier and sacrificial action. Even if damaged, the coating corrodes preferentially to preserve the underlying steel. Hot-dip galvanizing produces thick, durable coatings, while electroplating creates thinner layers with excellent coverage of complex shapes.
Conversion coatings chemically alter the metal surface. Passivation treatments on stainless steel remove free iron and enhance the chromium oxide layer. Anodizing aluminum artificially grows a thick aluminum oxide layer that provides excellent corrosion and wear resistance. Chromate conversion coatings, though facing environmental restrictions, deliver outstanding protection on aluminum and zinc.
Recent developments include graphene-based coatings, nanocomposite coatings, and sol-gel systems. These advanced materials offer improved barrier properties, self-healing capability, and environmental friendliness compared to traditional chromium-based systems. Smart coatings with embedded sensors can detect corrosion initiation and trigger protective responses.
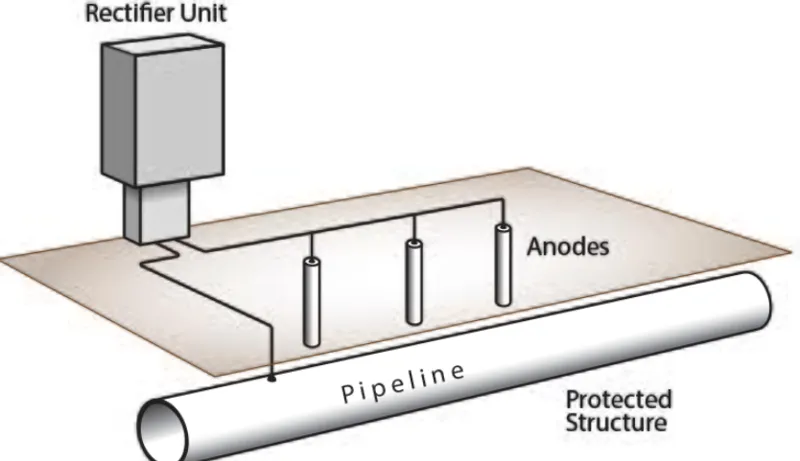
Cathodic Protection
Cathodic protection prevents corrosion by making the entire metal structure a cathode. Sacrificial anode systems connect more active metals (like zinc or magnesium) to the protected structure. The anode corrodes instead of the structure, providing protection as long as the anode remains effective.
Impressed current systems use an external power source to inject current, making the structure cathodic. This approach suits large structures like pipelines, ships, and offshore platforms. Proper design ensures current distributes uniformly to protect all exposed areas.
Both cathodic protection methods complement coatings. Even with protective coatings, small defects or damage can expose bare metal. Cathodic protection prevents corrosion at these defects, dramatically extending service life.
Corrosion Inhibitors
Chemical inhibitors reduce corrosion rates when added to the corrosive environment. Filming inhibitors adsorb onto metal surfaces, forming protective barriers. Passivating inhibitors promote oxide film formation on metal surfaces. Volatile corrosion inhibitors (VCIs) sublime to form protective vapor phases in enclosed spaces.
Inhibitors find use in cooling water systems, oil and gas production, concrete reinforcement protection, and temporary storage protection. Selection requires understanding of the corrosion mechanism, metal-electrolyte system, and compatibility with other chemicals present.
Green corrosion inhibitors derived from plant extracts and amino acids are replacing traditional chromate and phosphate inhibitors in many applications, addressing environmental and toxicity concerns.
Design Considerations
Proper design minimizes corrosion risk. Avoiding crevices, ensuring drainage to prevent water accumulation, and minimizing dissimilar metal contact all reduce corrosion. Rounded edges and smooth transitions eliminate stress concentrations that can initiate cracking.
Accessibility for inspection and maintenance allows early detection and treatment of corrosion. Designing for coating application and reapplication enables ongoing protection throughout the service life. Selecting materials appropriate for the environment from the beginning often proves more economical than extensive protection systems on unsuitable materials.

Industry-Specific Corrosion Resistance Requirements
Aerospace
Aircraft structures demand materials that combine high strength-to-weight ratios with adequate corrosion resistance. Aluminum alloys like 2024 and 7075 provide the required strength but need protective treatments. Aluminum cladding, anodizing, and protective primers prevent corrosion in service.
Stainless steels and titanium alloys serve in high-stress components, hydraulic systems, and engine parts. Regular inspection and protective treatments maintain corrosion resistance throughout the aircraft's operational life. Salt fog from coastal operations and de-icing chemicals create particularly aggressive environments.
Marine and Offshore
Seawater represents one of the most corrosive natural environments. Dissolved oxygen, salt content, temperature, and biological activity all contribute to rapid corrosion. Materials must resist general corrosion, pitting, crevice corrosion, and biofouling.
Copper-nickel alloys excel in seawater applications for piping, valves, and heat exchangers. Duplex and super duplex stainless steels provide strength with chloride resistance for structural components. Titanium's immunity to seawater corrosion makes it ideal for critical systems despite high cost.
Offshore oil and gas production combines seawater exposure with hydrogen sulfide, carbon dioxide, and high pressures. Corrosion-resistant alloys (CRAs) like 625, 825, and specialized stainless steels handle these extreme conditions. The global CRA market is growing at 8% annually, reaching an expected $16.27 billion by 2033, driven by demanding offshore applications.
Medical Devices
Biomedical applications require corrosion resistance in physiological environments-body fluids with chlorides, proteins, and pH variations. Materials must maintain mechanical properties while releasing no harmful corrosion products that could cause tissue reactions or implant failure.
Stainless steels like 316L serve in temporary implants and surgical instruments. Titanium and titanium alloys dominate permanent implants-joint replacements, bone plates, dental implants-due to their excellent corrosion resistance and biocompatibility. Cobalt-chromium alloys provide high wear resistance with good corrosion performance for articulating surfaces.
Surface treatments can enhance biocompatibility while maintaining corrosion resistance. Proper MIM processing enables complex medical components, but surface finishing and polishing are essential to achieve the corrosion resistance and biocompatibility required for body contact.
Chemical Processing
Chemical plants handle acids, bases, oxidizers, and organic solvents that aggressively attack most materials. Temperature and concentration affect corrosion rates dramatically-a material resistant to dilute acid at room temperature may fail rapidly in concentrated acid at elevated temperature.
Material selection relies on corrosion testing in specific process fluids. Nickel alloys like Hastelloy resist strong oxidizing acids. Zirconium and tantalum handle highly aggressive conditions. Glass-lined steel combines steel strength with glass's chemical inertness for reactors and vessels.
Trends in Corrosion Resistance
Research continues to develop more effective corrosion-resistant materials and protection methods. High-entropy alloys-complex compositions containing multiple principal elements-show promising corrosion resistance combined with excellent mechanical properties. These materials may outperform traditional alloys in extreme environments.
Nanostructured materials with refined grain sizes demonstrate improved corrosion resistance through more uniform protective oxide layer formation. Surface nanostructuring techniques can enhance existing materials without changing bulk composition.
AI and machine learning are revolutionizing corrosion prediction and monitoring. Smart sensors embedded in structures provide real-time corrosion monitoring, enabling predictive maintenance. AI analyzes sensor data to forecast when protective systems need replacement or when components approach failure.
Additive manufacturing enables creation of corrosion-resistant components with complex internal geometries impossible to produce conventionally. Tailoring composition locally within a part could optimize corrosion resistance where needed while using less expensive materials elsewhere.
Environmental pressures drive development of green corrosion protection technologies. Bio-based inhibitors, chromate-free conversion coatings, and recyclable protective systems reduce environmental impact while maintaining performance.
Frequently Asked Questions
What's the difference between corrosion resistance and rust resistance?
Rust specifically refers to iron oxide formation on iron-based metals like steel. Corrosion is the broader term for material degradation through chemical or electrochemical reactions, including rust but also oxidation of aluminum, copper, and other metals. A material can corrode without rusting if it doesn't contain iron.
Can corrosion-resistant materials ever corrode?
Yes, no material resists all corrosion in all environments. "Corrosion-resistant" indicates materials withstand specific conditions better than alternatives, not that they're completely immune. Even stainless steel can corrode in sufficiently aggressive environments or if its protective oxide layer is compromised. Proper material selection requires matching the material's resistance to the specific environment and application.
How do you improve the corrosion resistance of existing metal components?
Several approaches can enhance corrosion resistance after fabrication. Surface treatments like passivation, anodizing, or nitriding modify the surface chemistry. Protective coatings-paints, platings, or conversion coatings-create barriers. Cathodic protection systems use sacrificial anodes or impressed current to prevent corrosion. Corrosion inhibitors added to the environment reduce attack rates. The best method depends on the material, environment, and application constraints.
What factors should you consider when selecting corrosion-resistant materials?
Start with the specific corrosive environment-identify all chemicals, temperature ranges, and exposure conditions. Consider the required mechanical properties like strength, ductility, and fatigue resistance. Fabrication requirements affect material choice-some corrosion-resistant alloys are difficult to weld or machine. Cost matters but should factor in lifecycle expenses including maintenance and replacement, not just initial material price. Availability in required forms and sizes, and industry-specific regulations or certifications, also influence selection.














